Articles
- Page Path
- HOME > Epidemiol Health > Volume 44; 2022 > Article
-
Original Article
Non-linear association between serum folate concentrations and dyslipidemia: Korea National Health and Nutrition Examination Survey 2016-2018 -
Taiyue Jin1
 , Eun Young Park1
, Eun Young Park1 , Byungmi Kim1
, Byungmi Kim1 , Jin-Kyoung Oh1,2
, Jin-Kyoung Oh1,2
-
Epidemiol Health 2022;44:e2022046.
DOI: https://doi.org/10.4178/epih.e2022046
Published online: May 15, 2022
1Division of Cancer Prevention, National Cancer Control Institute, National Cancer Center, Goyang, Korea
2Department of Cancer Control and Population Health, Graduate School of Cancer Science and Policy, National Cancer Center, Goyang, Korea
- Correspondence: Eun Young Park Division of Cancer Prevention, National Cancer Control Institute, National Cancer Center, 323 Ilsan-ro, Ilsandong-gu, Goyang 10408, Korea E-mail: goajoa@ncc.re.kr
©2022, Korean Society of Epidemiology
This is an open-access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
OBJECTIVES
- We aimed to evaluate the association between serum folate concentrations and the prevalence of dyslipidemia.
-
METHODS
- A total of 4,477 adults (2,019 male and 2,458 female) enrolled in the Korea National Health and Nutrition Examination Survey (KNHANES) 2016-2018 were included. Serum samples were used to assess folate concentrations and total cholesterol (TC), triglyceride (TG), low-density lipoprotein (LDL)-cholesterol, and high-density lipoprotein (HDL)-cholesterol levels. Multivariate logistic regression with sampling weights was used to calculate odds ratios (ORs) and 95% confidence intervals (CIs).
-
RESULTS
- Elevated TC, TG, LDL-cholesterol and HDL-cholesterol levels were observed in 506 (11.3%), 646 (14.4%), 434 (9.7%), and 767 (17.1%) participants, respectively. We found non-linear trends between serum folate concentrations and the prevalence of hypercholesterolemia and hyper-LDL cholesterolemia from the restricted cubic smoothing spline. A higher prevalence of hypercholesterolemia was observed among participants in the first tertile of serum folate concentrations (OR,1.38; 95% CI, 1.05 to 1.79) than among those in the second tertile. However, a higher prevalence of hyper-LDL cholesterolemia was identified for both the first and third serum folate concentration tertiles (OR, 1.49; 95% CI, 1.08 to 2.05 and OR, 1.63; 95% CI, 1.20 to 2.20, respectively); furthermore, in these tertiles, the prevalence of hyper-LDL cholesterolemia was more pronounced among obese participants.
-
CONCLUSIONS
- Non-linear associations may exist between serum folate concentrations and the prevalence of hypercholesterolemia and hyper-LDL cholesterolemia in adults. The findings suggest that more accurate recommendations about folate intake and folic acid fortification and supplementation should be provided.
- Following the United Nations (UN) High-Level Meeting on the prevention and control of non-communicable diseases (NCDs) [1], the UN General Assembly adopted the goal to “by 2030, reduce by one third premature mortality from NCDs through prevention and treatment and promote mental health and well-being” [2]. Most NCD deaths are attributed to cardiovascular diseases (CVDs), including myocardial infarction (MI), ischemic heart disease, and stroke [3]. In 2019, around 17.9 million deaths worldwide were caused by CVDs, according to the World Health Organization (WHO) [4].
- Dyslipidemia is a well-known risk factor for CVDs. According to the 2019 WHO CVD risk charts, the 10-year risk of MI or coronary heart disease (CHD) is significantly increased in both male and female, by 26% and 23% per 1 mmol/L of the total cholesterol (TC) level, respectively [5]. Data from the WHO Global Health Observatory Data Repository showed that in 2008, the prevalence of hypercholesterolemia was highest in Europe (53.7%) and the Americas (47.7%), closely followed by the Western Pacific (36.7%) and South-East Asia (30.3%) [6]. In Korea particularly, from 2005 to 2018, the age-standardized prevalence of hypercholesterolemia increased from 8.0% to 21.4% among adults aged 30 years and over [7].
- Folate, an essential water-soluble nutrient belonging to the vitamin B family, is naturally present in a variety of foods such as green leafy vegetables, fruits, nuts, and beans [8]. Folic acid is the oxidized form of folate and is usually found in fortified foods and supplements. According to the Dietary Reference Intakes published by the Institute of Medicine, the recommended dietary allowance for folic acid fortification and supplementation among both adult male and female in the United States is 400 μg/day of dietary folate equivalents [9], which is the same as that for Korean adults [10].
- Previous studies have shown an inconsistent association between folate status and risk of CVDs [11-15]. A United States cohort study suggested that low serum folate concentrations were associated with increased CVD mortality [14]. However, several epidemiological studies and meta-analyses have reported that the risk of CVD incidence and mortality was not related to serum folate concentrations [12,13,15]. In relation to CHD, a United States cohort study reported that low serum folate concentrations were significantly associated with decreased risk of CHD in participants aged 55-77 years, but was associated with increased CHD risk in those aged 35-54 years [11]. Furthermore, only a few studies have evaluated whether cholesterol levels are modified by folate status; they have demonstrated inverse associations with levels of TC [16], triglyceride (TG) [16,17], and low-density lipoprotein (LDL)-cholesterol [18], and a positive association with high-density lipoprotein (HDL)-cholesterol levels [17,18].
- Given the unclear evidence, it is meaningful to evaluate the association between folate status and risk of dyslipidemia in the general population. In this cross-sectional study, we address the question of whether serum folate concentrations are associated with the prevalence of dyslipidemia using nationally representative data from the Korea National Health and Nutrition Examination Survey (KNHANES).
INTRODUCTION
- Study participants
- Participants in this study were recruited from the KNHANES, which is a complex, stratified, multi-stage probability sample survey conducted by the Korea Disease Control and Prevention Agency (KDCA). Annually, socio-demographic characteristics, lifestyle factors, anthropometric indices, biochemical profiles, nutritional status, and disease history are collected. For measurement of serum vitamins A, E, and folate, the participants were randomly extracted by survey district, sex, and age (in units of 5 years) from the whole survey population. Further information on the KNHANES can be found elsewhere [19].
- A total of 7,185 participants (3,288 male and 3,897 female) with serum folate measurements in the KNHANES 2016-2018 were included. Participants under 19 years of age (n=765) and pregnant female (n=20) were excluded. Participants were also excluded based on the following criteria: having been diagnosed by physicians or treated with medications for dyslipidemia (n=717), CVDs (stroke and MI) (n=120), or cancers (n=192); having an implausible level of total energy intake (± 3 standard deviations from the natural log-transformed mean) (n=32); and not providing information on body mass index (BMI) (n=7), alcohol consumption (n=43), smoking status (n=4), total energy intake (n=710), or menopausal status (n=86). Participants with a negative LDL-cholesterol value were also excluded (n=12). As a result, a total of 4,477 adults (2,019 male and 2,458 female) aged 19 years to 80 years were included in the final analysis. Figure 1 presents a flow diagram of the inclusion of study participants.
- Laboratory analysis
- Participants were asked to fast overnight before the blood draw. Serum folate concentration (ng/mL) was measured by the Chemiluminescent Microparticle Immunoassay (CIMA) method using Architect i4000SR (Abbott, Chicago, IL, USA). Serum TC (mg/dL) and TG (mg/dL) levels were determined by the enzymatic methods using the Hitachi Automatic Analyzer 7600-210 (Hitachi, Tokyo, Japan). The homogeneous enzymatic colorimetric method was used to assess the serum HDL-cholesterol level (mg/dL). Information on the laboratory data quality control program can be found elsewhere [20].
- Ascertainment of cases
- Serum LDL-cholesterol level (mg/dL) was estimated using the Friedewald formula [21]. In accordance with the Third Report of the Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (ATP III) by the National Cholesterol Education Program (NCEP) [22], dyslipidemia was defined as any of the following: (1) hypercholesterolemia with a TC level ≥ 240 mg/dL (6.22 mmol/L); (2) hypertriglyceridemia with a TG level ≥ 200 mg/dL (2.26 mmol/L); (3) hyper-LDL cholesterolemia with an LDL-cholesterol level ≥ 160 mg/dL (4.14 mmol/L); or (4) hypo-HDL cholesterolemia with an HDL-cholesterol level < 40 mg/dL (1.03 mmol/L).
- Covariates
- Body weight (kg) and height (m) were measured using a Giant-150N calibrated balance-beam scale (Hana, Seoul, Korea) and a portable stadiometer (Seriter, Bismarck, ND, USA), respectively. BMI (kg/m2) was defined as body weight divided by height squared. According to the WHO Western Pacific guideline, a BMI higher than 25 kg/m2 was defined as indicating obesity [23]. For past and current smokers, pack-years were calculated by multiplying the duration of smoking (year) by the number of cigarettes (packs/day). The frequency and average amount of any type of alcoholic beverage consumed were asked and used to calculate daily alcohol consumption (drinks/day). Type 2 diabetes and hypertension were defined based on self-reported diagnosis by physicians or treatment. The 24-hour dietary recall method was used to assess total energy intake (kcal/day) and dietary fiber intake (g/day).
- Statistical analysis
- Due to the skewed distribution, serum folate concentrations were natural log-transformed. The geometric means (GMs) and 95% confidence intervals (CIs) of serum folate concentrations were calculated according to sex.
- We examined whether non-linear associations existed between serum folate concentrations and dyslipidemia. A restricted cubic smoothing spline was constructed with five knots (10th, 25th, 50th, 75th, and 90th percentiles of natural log-transformed serum folate concentrations) using the R package “rms” (https://CRAN.R-project.org/package=rms). PROC SURVEYLOGISTIC was used to calculate the odds ratios (ORs) and 95% CIs. Serum folate concentrations were grouped into tertiles, and the second tertile was regarded as the reference group because participants with mid-range serum folate concentrations appeared to be minimally affected by dyslipidemia in the restricted cubic smoothing spline. The median concentration of serum folate was assigned to each group and used to test the linear trend. Moreover, we also conducted a subgroup analysis according to obesity status (< 25 kg/m2 or ≥ 25 kg/m2) by comparing the models with or without an interaction term using the likelihood ratio test. In the multivariable analyses, we adjusted for age (years, continuous), sex (for male and female combined), BMI (kg/m2, continuous), survey year (2016, 2017, or 2018), smoking status (pack-years, continuous), alcohol consumption (non-drinkers, < 1, 1, > 1 to 2, > 2 to 3, or > 3 drinks/day), menopausal status (for female, premenopausal or postmenopausal), type 2 diabetes (yes or no), hypertension (yes or no), and total energy intake (kcal/day, continuous). In addition, we also adjusted for dietary fiber intake (g/day, continuous) since the fermentation of dietary fiber may promote the microbial biosynthesis of folate by stimulating bacterial growth [24,25]. All analyses except the subgroup analysis were performed with sampling weights due to statistical intricacies. SAS version 9.4 (SAS Institute Inc., Cary, NC, USA) was used for all statistical analyses, and a p-value < 0.05 in 2-sided tests was defined as a significant difference.
- Ethics statement
- The study protocol was approved by the Institutional Review Board (IRB) of the KDCA (IRB No. 2018-01-03-PA). Informed consent was confirmed by the IRB.
MATERIALS AND METHODS
- Baseline characteristics
- Of the 4,477 participants, 506 (11.3%) had hypercholesterolemia, 646 (14.4%) had hypertriglyceridemia, 434 (9.7%) had hyper-LDL cholesterolemia, and 767 (17.1%) had hypo-HDL cholesterolemia. Participants with any type of dyslipidemia were older and had a higher BMI than those without dyslipidemia (Table 1). Female were more likely to have hypercholesterolemia and hyper-LDL cholesterolemia, whereas male were more likely to have hypertriglyceridemia and hypo-HDL cholesterolemia. All types of dyslipidemia had higher proportions of postmenopausal female than the corresponding group with findings in the acceptable range. Smokers and alcohol drinkers were more likely to have hypertriglyceridemia than their counterparts. Participants who were classified as having hypertriglyceridemia and hypo-HDL cholesterolemia had a higher total energy intake and a higher dietary fiber intake.
- Distribution of serum folate concentrations
- The overall GM of serum folate concentrations was 6.35 ng/mL (95% CI, 6.23 to 6.47) (Table 2). Female had higher serum folate concentrations than male (male: GM, 5.41 ng/mL; 95% CI, 5.27 to 5.55; female: GM, 7.47 ng/mL; 95% CI, 7.31 to 7.63).
- Association between serum folate concentrations and the prevalence of dyslipidemia
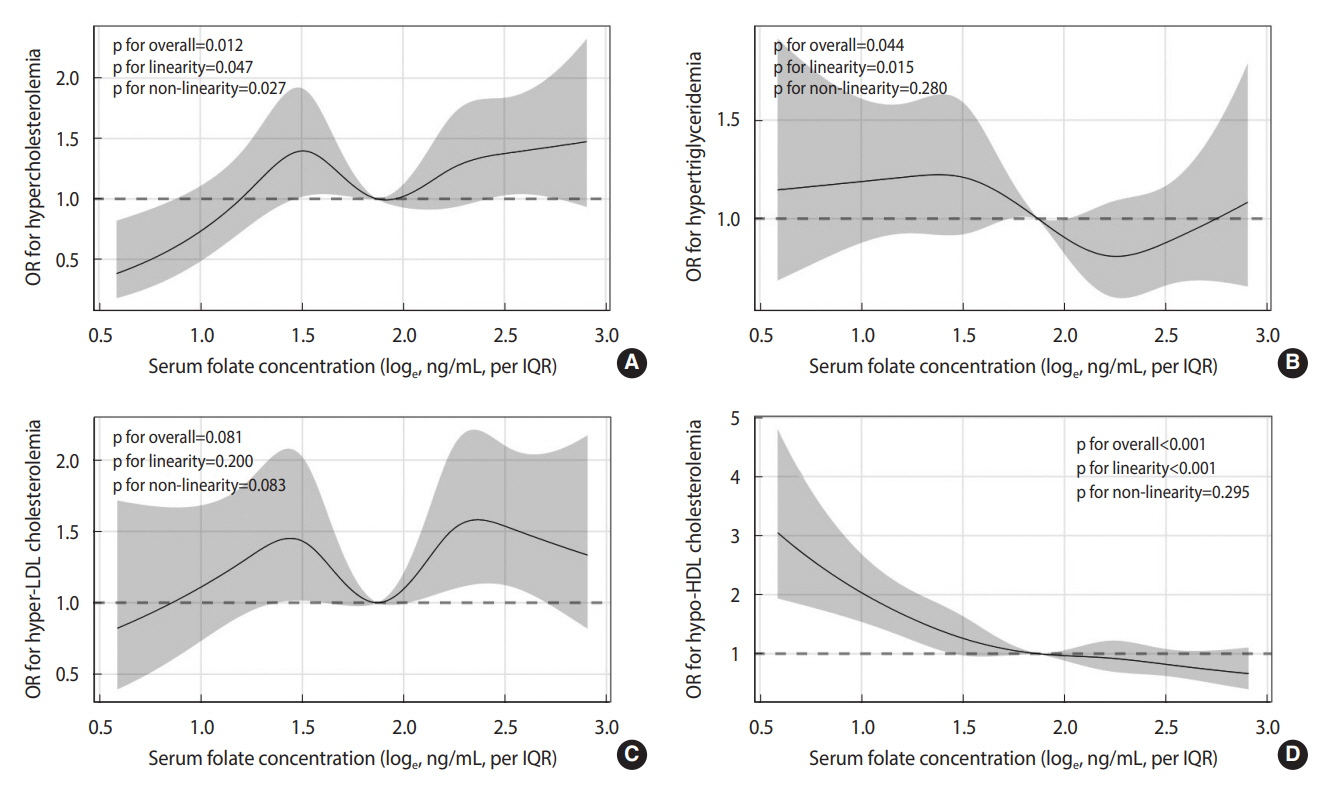
- With increasing serum folate concentrations, the OR for hypercholesterolemia showed a significant non-linearity (p for non-linearity= 0.027) (Figure 2A). Notably, although not statistically significant, there were tendencies for non-linear relationships between serum folate concentrations and the prevalence of hypertriglyceridemia and hyper-LDL cholesterolemia (p for non-linearity: 0.280 and 0.083, respectively) (Figure 2B and C). However, in contrast, the prevalence of hypo-HDL cholesterolemia linearly decreased with increments in serum folate concentrations (p for linearity < 0.001) (Figure 2D).
- When categorical models were applied, compared with participants in the second tertile, those in the third tertile had a higher prevalence of hypercholesterolemia (OR, 1.38; 95% CI, 1.05 to 1.79) (Table 3). This positive association was persistent only in female (third vs. second tertile: OR, 1.50; 95% CI, 1.07 to 2.09). However, in all the models, no significant linear trend was observed (p for trend: 0.228 for male and female combined, 0.761 for male, and 0.091 for female). After adjusting for potential confounding factors, the prevalence of hypertriglyceridemia was not associated with serum folate concentrations. For hyper-LDL cholesterolemia, a significant association with serum folate concentrations was observed in both the first and the third tertiles (OR, 1.49; 95% CI, 1.08 to 2.05 and OR, 1.63; 95% CI, 1.20 to 2.20, respectively), although the linear trend was not significant (p for trend=0.448). Additionally, female in the third tertile had an 83% higher prevalence of hyper-LDL cholesterolemia (95% CI, 1.27 to 2.65; p for trend=0.190). On the other hand, the prevalence of hypo-HDL cholesterolemia was inversely associated with serum folate concentrations among the first tertile participants compared to those in the second tertile (OR, 1.31; 95% CI, 1.02 to 1.69). Notably, there were significant linear trends in the association between serum folate concentrations and the prevalence of hypo-HDL cholesterolemia (p for trend < 0.001 for male and female combined, 0.005 for male, and 0.006 for female). When we further adjusted for dietary fiber intake, similar associations between serum folate concentrations and the prevalence of dyslipidemia were observed (Supplementary Material 1).
- Subgroup analysis according to obesity status
- As shown in Supplementary Material 2, associations between serum folate concentrations and the prevalence of hypercholesterolemia, hypertriglyceridemia, and hypo-HDL cholesterolemia were not modified by obesity status. However, the prevalence of hyper-LDL cholesterolemia was more pronounced among obese participants in both the first and the third tertiles than among those in the second tertile (OR, 1.72; 95% CI, 1.04 to 2.84 and OR, 2.15; 95% CI, 1.28 to 3.63, respectively), although the interaction was not statistically significant (p for interaction=0.702).
RESULTS
- In this study, we evaluated the association between serum folate concentrations and the prevalence of dyslipidemia among adults from a nationally representative dataset in Korea. The prevalence of hypercholesterolemia was non-linearly associated with serum folate concentrations, with a higher prevalence among participants with high concentrations of serum folate. Similarly, a non-linear association was found between serum folate concentrations and the prevalence of hyper-LDL cholesterolemia, and the prevalence was significantly higher among participants with low or high serum folate concentrations. In contrast, an inverse association between serum folate concentrations and the prevalence of hypo-HDL cholesterolemia was observed. Given the non-linear associations between serum folate concentrations and cholesterol levels, more accurate recommendations about folate intake and folic acid fortification and supplementation should be provided.
- In their 2006 Guidelines on Food Fortification with Micronutrients, the WHO and Food and Agricultural Organization (FAO) of the UN defined serum folate deficiency as less than 4.40 ng/mL (10 nmol/L) [26]. Since 1998, to prevent folate-related diseases, policies for folic acid fortification in food sources have been adopted in several counties. For example, folic acid has been fortified by 154 μg per 100 g of wheat flour in the United States, 150 μg in Canada, and 220 μg in Chile [26]. After fortification, serum folate concentrations increased in all 3 countries: from 5.50 ng/mL to 12.20 ng/mL (median) in the United States [27], from 5.94 ng/mL to 7.96 ng/mL (GM) in Canada [28], and from 4.27 ng/mL to 16.37 ng/mL (mean) in Chile [29]. In contrast, in countries where wheat flour has not been fortified with folic acid, the concentrations of folate are much lower: 7.92 ng/mL for male and 9.33 ng/mL for female (serum mean) in the United Kingdom [30], 6.91 ng/mL (plasma mean) in Spain [31], 7.35 ng/mL (plasma GM) in South China [32], and 8.20 ng/mL for male and 10.90 ng/mL for female (serum mean) in Taiwan [33]. However, folate concentrations in these non-fortified countries, including the Korean population in this study (6.35 ng/mL [serum GM]), are still higher than the cut-off level for folate deficiency suggested in the WHO and FAO guidelines.
- Only a few studies have reported the associations between folate status and cholesterol levels. In a German cross-sectional study, LDL-cholesterol levels significantly decreased by 0.164 mmol/L (6.34 mg/dL) per unit increment of plasma folate concentrations, whereas HDL-cholesterol levels increased by 0.094 mmol/L (3.64 mg/dL) [18]. Likewise, in the Chinese population, a positive association between serum folate concentrations and HDL-cholesterol levels has been observed [17]. Additionally, a recent meta-analysis of 34 randomized controlled trials (RCTs) demonstrated that individuals who received folic acid supplementation had significantly lower TC and TG levels (by 3.96 and 9.78 mg/dL, respectively) than those who received placebo [16]. In line with those findings, we identified a linear trend of decreasing hypo-HDL cholesterolemia prevalence with serum folate concentration increments. However, we also observed significant non-linear associations between serum folate concentrations and the prevalence of hypercholesterolemia and hyper-LDL cholesterolemia, with higher prevalence among individuals in the lower or higher serum folate concentration tertiles. The findings of this study need to be replicated in large prospective studies.
- The mechanisms of the adverse effects of folate deficiency have been widely investigated. Folate and folic acid in the diet are reduced to tetrahydrofolate (THF) within intestinal cells [34]. THF, which is one of the coenzyme forms of folate, accepts a methyl group from the catabolism of several amino acids (serine, glycine, sarcosine, etc.) [34,35]. The THF derivatives act as one-carbon donors for the remethylation of homocysteine, a well-known independent risk factor for CVDs [13,36]. Methionine generated by homocysteine remethylation is further involved in converting phosphatidylethanolamine (PE) to phosphatidylcholine (PC) [37,38], a major component of biological membranes, bile, and lipoproteins [39]. Folate deficiency reduces PC synthesis by inhibiting the methionine-homocysteine cycle, which further results in hepatic steatosis [40,41]. Additionally, PC synthesis is suppressed because folate deficiency also reduces the activity of the phosphatidylethanolamine N-methyltransferase, a transferase that converts PE to PC [35,39].
- We identified a significantly higher prevalence of hypercholesterolemia and hyper-LDL cholesterolemia among participants in the higher serum folate concentration tertile. There is increasing evidence for the adverse health effects of high folate concentrations. Unmetabolized folic acid (UMFA) is an indicator of excessive folate and folic acid intake [42,43] and in a clinical trial [44], UMFA has been found in participants supplied with 200 μg/meal or above of folic acid. Recent studies have also reported that UMFA may lead to the accumulation of dihydrofolate [43,45], the rate-limiting cofactor in converting folic acid to THF [46]. An in vivo study has suggested that excessive folic acid intake may also reduce methylenetetrahydrofolate reductase (MTHFR) protein levels in folic acid-supplemented diet-fed mice [42]. Decreased MTHFR levels are also linked to reduced homocysteine remethylation capacity, as MTHFR inhibits the conversion of THF to methyltetrahydrofolate [46,47]. Further prospective studies and RCTs are needed to clarify whether adverse effects of excessive folate and folic acid intake on cholesterol levels exist in the human population.
- In this study, the non-linear association between serum folate concentrations and the prevalence of hyper-LDL cholesterolemia was more pronounced among obese participants. An inverse association between serum folate concentrations and BMI has also been reported in previous epidemiological studies [48,49]. However, contrary to our results, a clinical trial found that TC and LDL-cholesterol levels significantly decreased among overweight and obese participants after 3-month supplementation with folic acid [50]. The limited evidence for the effect modification in the association between folate concentrations and the risk of dyslipidemia warrants further investigation.
- To our knowledge, we were the first to investigate the non-linear association between folate concentrations and dyslipidemia in the general population. The strengths of this study include the large sample size, adjustment for potential confounding factors, and generalizability. Furthermore, to minimize potential bias, we excluded participants who had been diagnosed with dyslipidemia, CVDs, or cancers before the baseline survey. However, our study had some limitations. Firstly, the findings from this cross-sectional study failed to elucidate a causal link between folate status and risk of dyslipidemia. Secondly, due to the lack of information on folic acid supplement use in the KNHANES, we could not evaluate whether the association between serum folate concentrations and the prevalence of dyslipidemia was modified by dietary folic acid supplementation. We also could not identify participants who supplemented with multivitamins, especially vitamins B12, B6, and B2, which are related to one-carbon metabolism [38]. However, to control for potential dietary confounders [25], we conducted a further analysis that adjusted for dietary fiber intake and found similar associations. Thirdly, despite all efforts to adjust for bias, residual confounding factors may have persisted.
- In conclusion, we found that serum folate concentrations were non-linearly associated with hypercholesterolemia and hyper-LDL cholesterolemia, with a higher prevalence among individuals with lower or higher serum folate concentrations. The findings suggest that more accurate recommendations on folate intake and folic acid fortification should be provided, and to prevent dyslipidemia in adults, public health strategies for maintaining adequate folate status should be implemented.
DISCUSSION
SUPPLEMENTARY MATERIALS
Supplementary Material 1.
Supplementary Material 2.
-
CONFLICT OF INTEREST
The authors have no conflicts of interest to declare for this study.
-
FUNDING
None.
-
AUTHOR CONTRIBUTIONS
Conceptualization: Park EY. Formal analysis: Jin T. Funding acquisition: None. Writing – original draft: Jin T. Writing – review & editing: Park EY, Kim B, Oh JK.
NOTES
ACKNOWLEDGEMENTS


| Variables | Concentration range (ng/mL) |
Overall |
Male |
Female |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Case/total | Crude | Adjusted1 | Case/total | Crude | Adjusted1 | Case/total | Crude | Adjusted1 | |||
| Hypercholesterolemia | |||||||||||
| Tertile 1 | 1.5-5.2 | 158/1,501 | 1.04 (0.79, 1.37) | 1.14 (0.84, 1.55) | 108/967 | 1.14 (0.77, 1.68) | 1.17 (0.77, 1.76) | 50/534 | 0.97 (0.63, 1.51) | 1.09 (0.68, 1.74) | |
| Tertile 2 | 5.3-8.2 | 155/1,509 | 1.00 (reference) | 1.00 (reference) | 60/642 | 1.00 (reference) | 1.00 (reference) | 95/867 | 1.00 (reference) | 1.00 (reference) | |
| Tertile 3 | 8.3-35.9 | 193/1,467 | 1.57 (1.22, 2.03) | 1.38 (1.05, 1.79) | 43/410 | 1.13 (0.72, 1.80) | 1.13 (0.71, 1.80) | 150/1,057 | 1.69 (1.22, 2.34) | 1.50 (1.07, 2.09) | |
| p for trend | 0.005 | 0.228 | 0.846 | 0.761 | 0.005 | 0.091 | |||||
| Hypertriglyceridemia | |||||||||||
| Tertile 1 | 1.5-5.2 | 283/1,501 | 1.28 (1.02, 1.61) | 1.05 (0.82, 1.35) | 218/967 | 0.99 (0.74, 1.32) | 0.99 (0.73, 1.34) | 65/534 | 1.35 (0.90, 2.02) | 1.28 (0.81, 2.02) | |
| Tertile 2 | 5.3-8.2 | 214/1,509 | 1.00 (reference) | 1.00 (reference) | 134/642 | 1.00 (reference) | 1.00 (reference) | 80/867 | 1.00 (reference) | 1.00 (reference) | |
| Tertile 3 | 8.3-35.9 | 149/1,467 | 0.74 (0.58, 0.95) | 0.85 (0.65, 1.11) | 77/410 | 0.94 (0.66, 1.35) | 0.94 (0.65, 1.37) | 72/1,057 | 0.82 (0.56, 1.21) | 0.80 (0.53, 1.21) | |
| p for trend | <0.001 | 0.178 | 0.811 | 0.835 | 0.037 | 0.076 | |||||
| Hyper-LDL cholesterolemia | |||||||||||
| Tertile 1 | 1.5-5.2 | 135/1,501 | 1.22 (0.90, 1.64) | 1.49 (1.08, 2.05) | 85/967 | 1.26 (0.83, 1.91) | 1.34 (0.87, 2.07) | 50/534 | 1.33 (0.85, 2.09) | 1.62 (1.00, 2.62) | |
| Tertile 2 | 5.3-8.2 | 119/1,509 | 1.00 (reference) | 1.00 (reference) | 48/642 | 1.00 (reference) | 1.00 (reference) | 71/867 | 1.00 (reference) | 1.00 (reference) | |
| Tertile 3 | 8.3-35.9 | 180/1,467 | 1.99 (1.48, 2.68) | 1.63 (1.20, 2.20) | 39/410 | 1.37 (0.80, 2.36) | 1.26 (0.72, 2.19) | 141/1,057 | 2.13 (1.49, 3.04) | 1.83 (1.27, 2.65) | |
| p for trend | 0.002 | 0.448 | 0.975 | 0.613 | 0.005 | 0.190 | |||||
| Hypo-HDL cholesterolemia | |||||||||||
| Tertile 1 | 1.5-5.2 | 353/1,501 | 1.54 (1.23, 1.94) | 1.31 (1.02, 1.69) | 285/967 | 1.28 (0.96, 1.70) | 1.35 (1.00, 1.82) | 68/534 | 1.22 (0.82, 1.83) | 1.23 (0.78, 1.93) | |
| Tertile 2 | 5.3-8.2 | 238/1,509 | 1.00 (reference) | 1.00 (reference) | 152/642 | 1.00 (reference) | 1.00 (reference) | 86/867 | 1.00 (reference) | 1.00 (reference) | |
| Tertile 3 | 8.3-35.9 | 176/1,467 | 0.71 (0.55, 0.92) | 0.77 (0.59, 1.01) | 93/410 | 0.96 (0.68, 1.35) | 0.85 (0.59, 1.21) | 83/1,057 | 0.75 (0.50, 1.10) | 0.68 (0.45, 1.02) | |
| p for trend | <0.001 | <0.001 | 0.046 | 0.005 | 0.019 | 0.006 | |||||
Values are presented as odds ratio (95% confidence interval).
LDL, low-density lipoprotein; HDL, high-density lipoprotein.
1 Multivariate logistic regression model adjusted for age (years, continuous), sex (for male and female combined), body mass index (kg/m2, continuous), survey year (2016, 2017, or 2018), smoking status (pack-year, continuous), alcohol consumption (non-drinkers, <1, 1, >1 to 2, >2 to 3, or >3 drinks/day), menopausal status (for female, premenopausal or postmenopausal), type 2 diabetes (yes or no), hypertension (yes or no), and total energy intake (kcal/day, continuous).
- 1. United Nations. Political declaration of the third high-level meeting of the General Assembly on the prevention and control of non-communicable diseases; 2018 [cited 2021 Oct 5]. Available from: https://digitallibrary.un.org/record/1648984.
- 2. United Nations. Goal 3: ensure healthy lives and promote well-being for all at all ages. [cited 2021 Oct 5]. Available from: https://www.un.org/sustainabledevelopment/health/.Article
- 3. World Health Organization. Noncommunicable diseases; 2021 [cited 2021 Oct 5]. Available from: https://www.who.int/news-room/fact-sheets/detail/noncommunicable-diseases.
- 4. . Cardiovascular diseases (CVDs); 2021 [cited 2021 Oct 5]. Available from: https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds).ArticlePDF
- 5. WHO CVD Risk Chart Working Group. World Health Organization cardiovascular disease risk charts: revised models to estimate risk in 21 global regions. Lancet Glob Health 2019;7:e1332-e1345.PubMedPMC
- 6. WHO CVD Risk Chart Working Group. Global health observatory data repository. [cited 2021 Oct 5]. Available from: https://apps.who.int/gho/data/view.main.2570?lang%C2%BCen.
- 7. Kim Y, Nho SJ, Woo G, Kim H, Park S, Kim Y, et al. Trends in the prevalence and management of major metabolic risk factors for chronic disease over 20 years: findings from the 1998-2018 Korea National Health and Nutrition Examination Survey. Epidemiol Health 2021;43:e2021028.ArticlePubMedPMC
- 8. Bailey LB, Stover PJ, McNulty H, Fenech MF, Gregory JF 3rd, Mills JL, et al. Biomarkers of nutrition for development-folate review. J Nutr 2015;145:1636S-1680S.ArticlePubMedPMC
- 9. Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate, Other B Vitamins, and Choline. Dietary reference intakes for thiamin, riboflavin, niacin, vitamin B6, folate, vitamin B12, pantothenic acid, biotin, and choline. Washington, DC: National Academies Press; 1998. p 196.
- 10. Ministry of Health and Welfare; Korean Nutrition Society. Dietary reference intakes for Koreans 2020. Sejong: Ministry of Health and Welfare; 2020. p 21 (Korean).
- 11. Giles WH, Kittner SJ, Croft JB, Anda RF, Casper ML, Ford ES. Serum folate and risk for coronary heart disease: results from a cohort of US adults. Ann Epidemiol 1998;8:490-496.PubMed
- 12. Bazzano LA, Reynolds K, Holder KN, He J. Effect of folic acid supplementation on risk of cardiovascular diseases: a meta-analysis of randomized controlled trials. JAMA 2006;296:2720-2726.ArticlePubMed
- 13. Miller ER 3rd, Juraschek S, Pastor-Barriuso R, Bazzano LA, Appel LJ, Guallar E. Meta-analysis of folic acid supplementation trials on risk of cardiovascular disease and risk interaction with baseline homocysteine levels. Am J Cardiol 2010;106:517-527.ArticlePubMed
- 14. Peng Y, Dong B, Wang Z. Serum folate concentrations and allcause, cardiovascular disease and cancer mortality: a cohort study based on 1999-2010 National Health and Nutrition Examination Survey (NHANES). Int J Cardiol 2016;219:136-142.ArticlePubMed
- 15. Kim HN, Eun YM, Song SW. Serum folate and vitamin B12 levels are not associated with the incidence risk of atherosclerotic events over 12 years: the Korean Genome and Epidemiology Study. Nutr Res 2019;63:34-41.ArticlePubMed
- 16. Asbaghi O, Ashtary-Larky D, Bagheri R, Nazarian B, Pourmirzaei Olyaei H, Rezaei Kelishadi M, et al. Beneficial effects of folic acid supplementation on lipid markers in adults: a GRADE-assessed systematic review and dose-response meta-analysis of data from 21,787 participants in 34 randomized controlled trials. Crit Rev Food Sci Nutr 2021;1-19.Article
- 17. Li WX, Lv WW, Dai SX, Pan ML, Huang JF. Joint associations of folate, homocysteine and MTHFR, MTR and MTRR gene polymorphisms with dyslipidemia in a Chinese hypertensive population: a cross-sectional study. Lipids Health Dis 2015;14:101.ArticlePubMedPMCPDF
- 18. Semmler A, Moskau S, Grigull A, Farmand S, Klockgether T, Smulders Y, et al. Plasma folate levels are associated with the lipoprotein profile: a retrospective database analysis. Nutr J 2010;9:31.ArticlePubMedPMCPDF
- 19. Kweon S, Kim Y, Jang MJ, Kim Y, Kim K, Choi S, et al. Data resource profile: the Korea National Health and Nutrition Examination Survey (KNHANES). Int J Epidemiol 2014;43:69-77.ArticlePubMedPMC
- 20. Kim Y. The Korea National Health and Nutrition Examination Survey (KNHANES): current status and challenges. Epidemiol Health 2014;36:e2014002.ArticlePubMedPMC
- 21. Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 1972;18:499-502.ArticlePubMedPDF
- 22. Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the third report of the national cholesterol education program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA 2001;285:2486-2497.ArticlePubMed
- 23. World Health Organization, Regional Office for the Western Pacific. The Asia-Pacific perspective: redefining obesity and its treatment; 2000 [cited 2021 Oct 5]. Available from: https://apps.who.int/iris/handle/10665/206936.
- 24. Houghton LA, Green TJ, Donovan UM, Gibson RS, Stephen AM, O’Connor DL. Association between dietary fiber intake and the folate status of a group of female adolescents. Am J Clin Nutr 1997;66:1414-1421.ArticlePubMed
- 25. Bingham SA, Norat T, Moskal A, Ferrari P, Slimani N, ClavelChapelon F, et al. Is the association with fiber from foods in colorectal cancer confounded by folate intake? Cancer Epidemiol Biomarkers Prev 2005;14:1552-1556.ArticlePubMedPDF
- 26. World Health Organization. Guidelines on food fortification with micronutrients; 2006 [cited 2021 Oct 5]. Available from: https://www.who.int/publications/i/item/9241594012.
- 27. McDowell MA, Lacher DA, Pfeiffer CM, Mulinare J, Picciano MF, Rader JI, et al. NCHS Data Brief. Cancer Epidemiol Biomarkers Prev. 2008. 1-8.PubMed
- 28. Liu S, West R, Randell E, Longerich L, O’connor KS, Scott H, et al. A comprehensive evaluation of food fortification with folic acid for the primary prevention of neural tube defects. BMC Pregnancy Childbirth 2004;4:20.ArticlePubMedPMCPDF
- 29. Hertrampf E, Cortés F. Folic acid fortification of wheat flour: Chile. Nutr Rev 2004;62(Suppl 1):S44-S48.ArticlePubMed
- 30. Bates B, Page P, Cos L, Nicholson S, Roberts C, Collins D, et al. National Diet and Nutrition Survey Rolling Programme (NDNS RP): supplementary report: blood folate results for the UK as a whole, Scotland, Northern Ireland (years 1 to 4 combined) and Wales (years 2 to 5 combined); 2017 [cited 2021 Oct 5]. Available from: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/661932/National_Diet_and_Nutrition_Survey_Rolling_Programme__NDNS_RP_.pdf.
- 31. Planells E, Sánchez C, Montellano MA, Mataix J, Llopis J. Vitamins B6 and B12 and folate status in an adult Mediterranean population. Eur J Clin Nutr 2003;57:777-785.ArticlePubMedPDF
- 32. Hao L, Ma J, Stampfer MJ, Ren A, Tian Y, Tang Y, et al. Geographical, seasonal and gender differences in folate status among Chinese adults. J Nutr 2003;133:3630-3635.ArticlePubMed
- 33. Chen KJ, Pan WH, Lin YC, Lin BF. Trends in folate status in the Taiwanese population aged 19 years and older from the Nutrition and Health Survey in Taiwan 1993-1996 to 2005-2008. Asia Pac J Clin Nutr 2011;20:275-282.PubMed
- 34. Zhao R, Matherly LH, Goldman ID. Membrane transporters and folate homeostasis: intestinal absorption and transport into systemic compartments and tissues. Expert Rev Mol Med 2009;11:e4.ArticlePubMedPMC
- 35. da Silva RP, Kelly KB, Al Rajabi A, Jacobs RL. Novel insights on interactions between folate and lipid metabolism. Biofactors 2014;40:277-283.ArticlePubMedPDF
- 36. Wierzbicki AS. Homocysteine and cardiovascular disease: a review of the evidence. Diab Vasc Dis Res 2007;4:143-150.ArticlePubMedPDF
- 37. Fox JT, Stover PJ. Folate-mediated one-carbon metabolism. Vitam Horm 2008;79:1-44.PubMed
- 38. Ducker GS, Rabinowitz JD. One-carbon metabolism in health and disease. Cell Metab 2017;25:27-42.ArticlePubMed
- 39. Cole LK, Vance JE, Vance DE. Phosphatidylcholine biosynthesis and lipoprotein metabolism. Biochim Biophys Acta 2012;1821:754-761.ArticlePubMed
- 40. Yao ZM, Vance DE. The active synthesis of phosphatidylcholine is required for very low density lipoprotein secretion from rat hepatocytes. J Biol Chem 1988;263:2998-3004.ArticlePubMed
- 41. Williams KT, Schalinske KL. Homocysteine metabolism and its relation to health and disease. Biofactors 2010;36:19-24.ArticlePubMed
- 42. Christensen KE, Mikael LG, Leung KY, Lévesque N, Deng L, Wu Q, et al. High folic acid consumption leads to pseudo-MTHFR deficiency, altered lipid metabolism, and liver injury in mice. Am J Clin Nutr 2015;101:646-658.ArticlePubMedPMC
- 43. Selhub J, Rosenberg IH. Excessive folic acid intake and relation to adverse health outcome. Biochimie 2016;126:71-78.ArticlePubMed
- 44. Kelly P, McPartlin J, Goggins M, Weir DG, Scott JM. Unmetabolized folic acid in serum: acute studies in subjects consuming fortified food and supplements. Am J Clin Nutr 1997;65:1790-1795.ArticlePubMed
- 45. Bailey SW, Ayling JE. The extremely slow and variable activity of dihydrofolate reductase in human liver and its implications for high folic acid intake. Proc Natl Acad Sci U S A 2009;106:15424-15429.ArticlePubMedPMC
- 46. Pietrzik K, Bailey L, Shane B. Folic acid and L-5-methyltetrahydrofolate: comparison of clinical pharmacokinetics and pharmacodynamics. Clin Pharmacokinet 2010;49:535-548.PubMed
- 47. Kalmbach RD, Choumenkovitch SF, Troen AP, Jacques PF, D’Agostino R, Selhub J. A 19-base pair deletion polymorphism in dihydrofolate reductase is associated with increased unmetabolized folic acid in plasma and decreased red blood cell folate. J Nutr 2008;138:2323-2327.ArticlePubMedPMC
- 48. Mahabir S, Ettinger S, Johnson L, Baer DJ, Clevidence BA, Hartman TJ, et al. Measures of adiposity and body fat distribution in relation to serum folate levels in postmenopausal women in a feeding study. Eur J Clin Nutr 2008;62:644-650.ArticlePubMedPDF
- 49. Tungtrongchitr R, Pongpaew P, Tongboonchoo C, Vudhivai N, Changbumrung S, Tungtrongchitr A, et al. Serum homocysteine, B12 and folic acid concentration in Thai overweight and obese subjects. Int J Vitam Nutr Res 2003;73:8-14.ArticlePubMed
- 50. Mierzecki A, Kłoda K, Bukowska H, Chełstowski K, Makarewicz-Wujec M, Kozłowska-Wojciechowska M. Association between low-dose folic acid supplementation and blood lipids concentrations in male and female subjects with atherosclerosis risk factors. Med Sci Monit 2013;19:733-739.ArticlePubMedPMC
REFERENCES
Figure & Data
References
Citations

- Association between traditional Korean fermented vegetables (kimchi) intake and serum lipid profile: using the Korean Genome and Epidemiology Study (KoGES) cohort
Seok-Jae Oh, Wooje Lee, Sung Wook Hong, Sangah Shin
European Journal of Nutrition.2024;[Epub] CrossRef - A Review of Major Secondary Data Resources Used for Research in Traditional Korean Medicine
Chunhoo Cheon, Bo-Hyoung Jang, Seong-Gyu Ko
Perspectives on Integrative Medicine.2023; 2(2): 77. CrossRef - The Utilization of Serum Folate and Homocysteine Tests and the Prevalence of Folate Deficiency in Reproductive-Age Korean Women during the COVID-19 Pandemic
Rihwa Choi, Wonseo Park, Gayoung Chun, Sang Gon Lee, Eun Hee Lee
Nutrients.2023; 15(14): 3236. CrossRef
- Figure
- Related articles
-
- Development and validation of the Health Literacy Index for the Community (HLIC) for the Korean National Health and Nutrition and Examination Survey
- Association of healthy lifestyle factors with the risk of hypertension, dyslipidemia, and their comorbidity in Korea: results from the Korea National Health and Nutrition Examination Survey 2019-2021
- Changes in food sufficiency among Korean adults in urban and rural areas during the COVID-19 pandemic: an analysis of the 7th and 8th Korea National Health and Nutrition Examination Survey
- Associations of the magnesium depletion score and magnesium intake with diabetes among US adults: an analysis of the National Health and Nutrition Examination Survey 2011-2018
- Folate, vitamin B12, and homocysteine status in the Korean population: data from the 2013-2015 Korea National Health and Nutrition Examination Survey

 KSE
KSE
 PubReader
PubReader ePub Link
ePub Link Cite
Cite




