Articles
- Page Path
- HOME > Epidemiol Health > Volume 37; 2015 > Article
-
Brief Communication
Epidemiological evidences on overdiagnosis of prostate and kidney cancers in Korean - Jong-Myon Bae
-
Epidemiol Health 2015;37:e2015015.
DOI: https://doi.org/10.4178/epih/e2015015
Published online: March 7, 2015
Department of Preventive Medicine, Jeju National University School of Medicine, Jeju, Korea
- Correspondence: Jong-Myon Bae Department of Preventive Medicine, Jeju National University School of Medicine, 102 Jejudaehak-ro, Jeju 690-756, Korea Tel: +82-64-755-5567, Fax: +82-64-725-2593, E-mail: jmbae@jejunu.ac.kr
• Received: February 2, 2015 • Accepted: March 7, 2015
©2015, Korean Society of Epidemiology
This is an open-access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/3.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
OBJECTIVES:
- The prostate specific antigen test is widely used as the main method of screening prostate cancer in Korea. Additionally, the use of ultrasound sonography may lead to overdiagnosis of kidney cancer as well as thyroid cancer. This study aimed to highlight epidemiological evidences regarding overdiagnosis of prostate and kidney cancers in Korean.
-
METHODS:
- The annual trends of national incidence and mortality of prostate and kidney cancers provided by the Korean Statistical Information Service were evaluated.
-
RESULTS:
- The rate of increase in the incidence of prostate and kidney cancer was 6 and 5 times higher than that of mortality between 2000 and 2011, respectively. Additionally, the age group showing the highest incidence in prostate cancer shifted from 85 years and older to 75-79 years.
-
CONCLUSIONS:
- This evidence suggests that prostate and kidney cancers are overdiagnosed in Korea. Further research in this area, using national cancer registry databases, should be encouraged to prevent overdiagnosis.
- Cancer screening, which reduces cancer mortality through early detection, inevitably leads to the problem of overdiagnosis [1]. Overdiagnosis is defined as ‘detection of cancers that would never have been found were it not for the screening test’ [2], and is regarded as a harmful aspect of screening testing because it results in unnecessary overtreatment [3]. In South Korea (hereafter Korea), concerns have recently been raised regarding the overdiagnosis of thyroid [4] and breast cancers [5].
- A debate on overdiagnosis occurs when a cancer screening project is introduced, or new screening tests for early detection of cancer are developed [2]. In the West, there have been claims of overdiagnosis relating to thyroid, breast, prostate, and kidney cancers [1]; however, in Korea, where testing for prostate specific antigen (PSA) levels in blood is universal, an investigation of overdiagnosis of prostate cancer would provide important evidence for cancer management policies.
- In addition, thyroid cancer shows the highest incidence among cancers in the Korean population [6], leading to claims of overdiagnosis due to expansion and supply of ultrasound testing devices [4]. However, ultrasound devices can increase the likelihood of discovering incidental cancer in the neck and in the abdominal organs [7]. Thus, one might suspect that kidney cancer is also currently being overdiagnosed. With respect to these questions, author aimed to investigate the epidemiological evidences on overdiagnosis of prostate and kidney cancers in Koreans.
INTRODUCTION
- The judgement criterion for the presence of overdiagnosis is that mortality does not change substantially compared to an in crease in incidence [2]. The annual trends of national incidence and mortality of prostate and kidney cancers provided by the Korean Statistical Information Service (KOSIS) [8] were evaluated.
MATERIALS AND METHODS
- Prostate cancer
- Welch and Black [1] presented evidence for prostate cancer overdiagnosis in that the incidence increased considerably immediately after an introduction of PSA screening; however, there was little change in mortality. There is similar evidence in relation to the overdiagnosis of thyroid cancer in Korea [7]. Figure 1(A) shows the annual incidence and mortality of prostate cancer provided by the KOSIS [8]. Compared with 2000, mortality in 2011 had increased by 3.3 (=5.6-2.3) deaths per 100,000 people, while prevalence increased by 20.4 (=27.7-7.3) cases per 100,000 people—a 6-fold increase (=20.4/ 3.3). This is the first piece of epidemiological evidences on the overdiagnosis of prostate cancer.
- At the same time, Hilton et al. [9] reported that the age of diagnosis had decreased since the expansion of PSA testing. Accordingly, Figure 1(B) describes the incidence for different age groups of Korean males over the last 10 years. The age group with the highest incidence shows a shifting trend, from the over-85 group in 2001, to the 80 to 84 years group from 2003 to 2009, and the 75 to 79 years group in 2011. This is the second piece of epidemiological evidences on overdiagnosis. However, for the evidence to be more convincing, it is necessary to determine whether there has been a change in the distribution of disease stage at diagnosis [10].
- Kidney cancer
- Approximately 10% of kidney cancers were incidental cancers in the early 1970s; however, with the development of various imaging technologies such as computer tomography and magnetic resonance imaging, this increased to approximately 60% in the 1990s [7]. Accordingly, the majority of kidney cancers are discovered incidentally while performing radiological tests for other purposes [11]. Compared with symptomatic tumors, incidental tumors are smaller, have favorable prognostic scores, and are less likely to metastasize [12]. As more incidental cancers are diagnosed, the incidence of kidney cancer will increase. Meanwhile, mortality is not expected to change, and even if it increases, the magnitude of change is not expected to be as large as that for incidence. This line of reasoning can explain the phenomenon in developed countries with a high economic level whereby the incidence of kidney cancer is high but mortality is low [13].
- With the rate of detection of incidental cancer increasing following the proliferation of radiodiagnostic machines, any large discrepancy between an increase in incidence and increase in mortality provides epidemiological evidence of overdiagnosis [1]. While mortality in kidney cancer doubled in the US between 1971 and 2010, incidence increased 5-fold [14]. In particular, a report showing a significant change in incidence supports a conclusion of overdiagnosis due to changes in the healthcare environment rather than in relation to age group or birth cohort over time [15]. Figure 2A shows the incidence and mortality trends of kidney cancer from 2000 to 2011 provided by the KOSIS [8]. Compared with 2000, mortality in 2011 had increased by 1.5-fold (=1.7/1.1), while incidence had doubled (=6.0/3.0); thus, there appears to be little difference. However, as mortality increased by 0.6 (=1.7-1.1) deaths per 100,000 people and incidence increased by 3.0 (=6.0-3.0) cases per 100,000 people, the rate of increase was 5 times (=3.0/0.6) higher for incidence compared with mortality. In other words, based on the difference in the slope of the curve in Figure 2, the gap between incidence and mortality is increasing with time to date.
- Meanwhile, one of epidemiological characteristics of kidney cancer is that it is twice as common in males compared with females [16]. Figure 2B presents the pattern of increase for incidence and mortality in females. Over the same time-period, the rate of increase for incidence was 2.2 times (=2/0.9) higher than mortality for males, and 6.0 times (=1.8/0.3) higher for females. Given that thyroid cancer, as detected by neck ultrasound, is far higher in females [6], future research will be necessary to investigate whether the larger discrepancy shown for females with kidney cancer is related to the diffusion of ultrasound.
- Esophageal cancer
- As mentioned above, if overdiagnosis of prostate cancer and kidney cancer results from expanded testing, other cancers that are the target of screening might also be overdiagnosed. In terms of national cancer screening, upper endoscopy for gastric cancer and mammography for breast cancer, at 70.9% each, are the most common clinical examinations [17]. With regards to upper endoscopy, the most commonly used method in screening, is it possible that esophageal cancer could be overdiagnosed given that the process involves transit through the esophagus to the stomach?
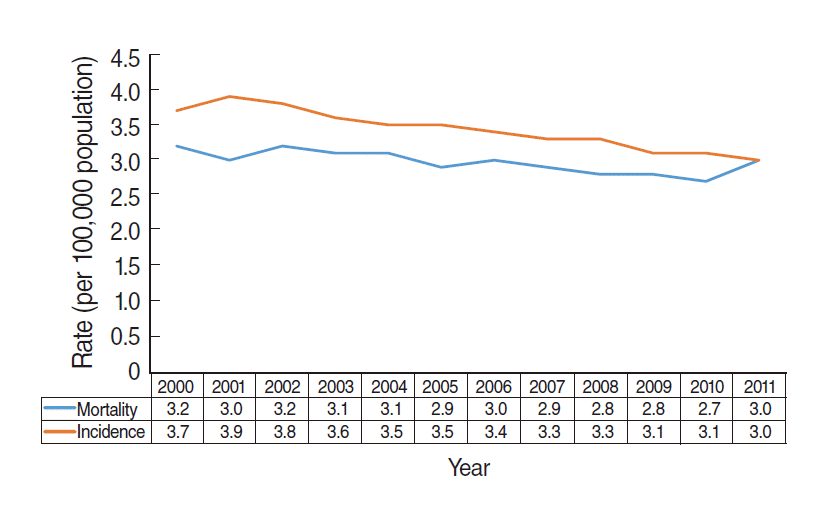
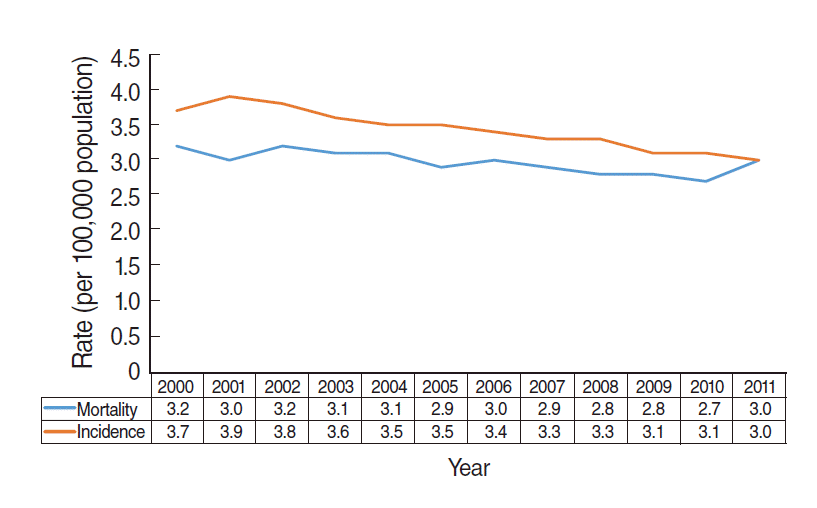
- Figure 3 shows the annual incidence and mortality for esophageal cancer provided by the KOSIS [8]. Between 2000 and 2011, both incidence and mortality decreased or were unchanged. These data demonstrate that there was no overdiagnosis of esophageal cancer, despite the performance of upper endoscopies. Accordingly, overdiagnosis occurs when a ‘length time bias’ is introduced due to the expansion of screening programmes for indolent cancers [2,3].
RESULTS
- There is a need for caution when interpreting the phenomenon of incidence-mortality discrepancy given as epidemiological evidences of overdiagnosis in Figure 1(A) and Figure 2(A). The reason, in terms of prostate cancer, can be explained by increased screening provision, improved diagnostic sensitivity, and changes in risk factors [19]. The discrepancy for kidney cancer can also be explained by increased testing, development of new screening methods such as genetic testing, changes in terms of risk factors such as quitting smoking, development of effective treatments, and medical care provision systems [16,18].
- Given the volume of epidemiological evidence relating to overdiagnosis, natural history studies of the pertinent cancers are required to determine its extent [2,16]. In other words, there is a need to calculate the scale of overdiagnosis in order to evaluate the risk-benefit payoff of screening [2]. Also, evidences relating to the phenomenon of the smaller size and lower stage of cancer at diagnosis in cases of overdiagnosis should be provided in the national cancer registration data managed by the Korea Central Cancer Registry (KCCR) [2,9]. With research limitations due to the reinforcement of personal information protection [20], author recommends the KCCR to take the lead in using the cancer registration data, constructed with the purpose of calculating national cancer statistics, to produce useful data for the development of cancer management policies. In addition, multifaceted research activities by healthcare workers to minimize overdiagnosis are also necessary [2].
DISCUSSION
ACKNOWLEDGEMENTS
SUPPLEMENTARY MATERIAL
Figure 1.Prostate cancer in Korean men. (A) Age-adjusted incidence and mortality between 2000 and 2011. (B) Age-adjusted incidence in 2001, 2003, 2005, 2007, 2009, and 2011 by age group.
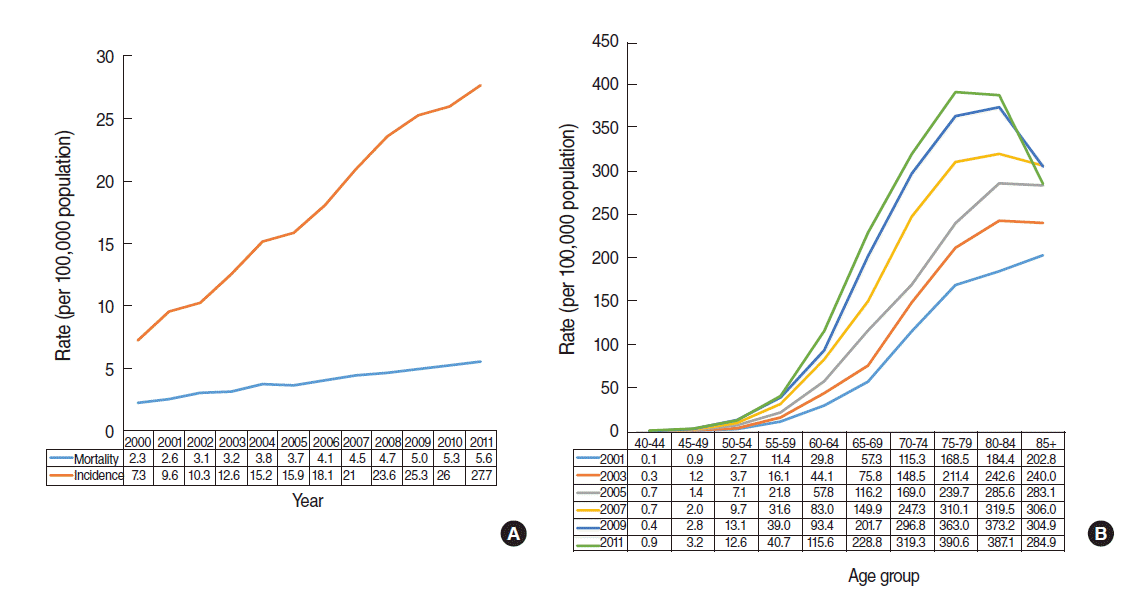

Figure 2.Age-adjusted incidence and mortality of kidney cancer between 2000 and 2011 in (A) all Koreans and (B) in Korean women.
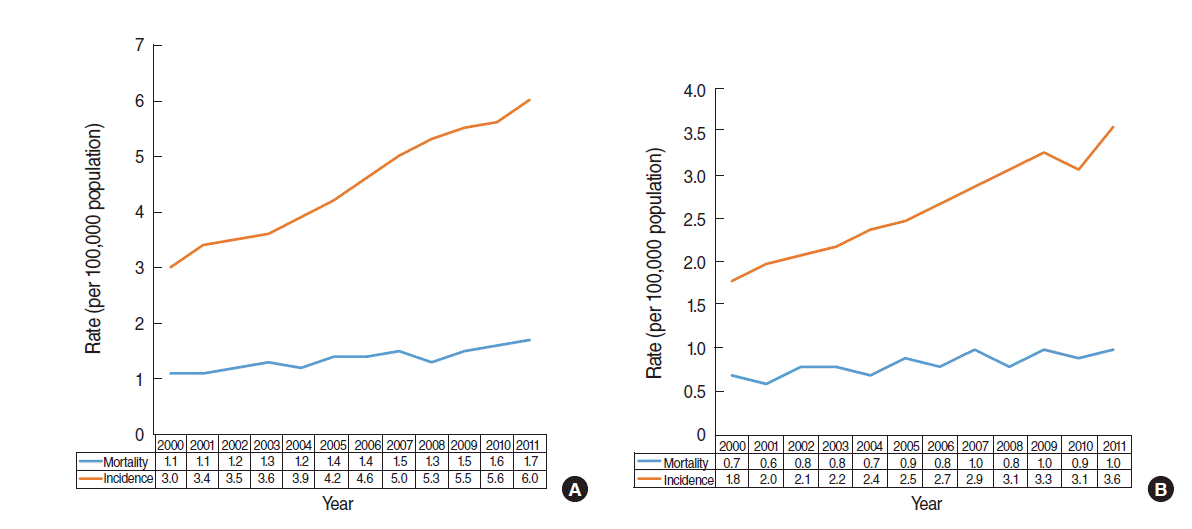

Figure 3.Age-adjusted incidence and mortality of esophageal cancer between 2000 and 2011 in the Korean population.

- 1. Welch HG, Black WC. Overdiagnosis in cancer. J Natl Cancer Inst 2010;102:605-613.ArticlePubMed
- 2. Bae JM. Overdiagnosis: epidemiologic concepts and estimation. Epidemiol Health 2015;37:e2015004.ArticlePubMedPMC
- 3. Dahm P, Neuberger M, Ilic D. Screening for prostate cancer: shaping the debate on benefits and harms. Cochrane Database Syst Rev 2013;9:ED000067.Article
- 4. Ahn HS, Kim HJ, Welch HG. Korea’s thyroid-cancer “epidemic”-screening and overdiagnosis. N Engl J Med 2014;371:1765-1767.ArticlePubMed
- 5. Bae JM. On the benefits and harms of mammography for breast cancer screening in Korean women. Korean J Fam Pract 2014;1:1-6 (Korean).
- 6. Jung KW, Won YJ, Kong HJ, Oh CM, Lee DH, Lee JS. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2011. Cancer Res Treat 2014;46:109-123.ArticlePubMedPMCPDF
- 7. Ng CS, Wood CG, Silverman PM, Tannir NM, Tamboli P, Sandler CM. Renal cell carcinoma: diagnosis, staging, and surveillance. AJR Am J Roentgenol 2008;191:1220-1232.ArticlePubMed
- 8. Korean Statistical Information Service. National cancer statistics. [cited 2015 Jan 30]. Available from: http://kosis.kr/statisticsList/statisticsList_01List.jsp?vwcd=MT_ZTITLE&parentId=A#SubCont (Korean).
- 9. Hilton WM, Padalecki SS, Ankerst DP, Leach RJ, Thompson IM. Temporal changes in the clinical approach to diagnosing prostate cancer. J Natl Cancer Inst Monogr 2012;2012:162-168.ArticlePubMedPMC
- 10. Li J, German R, King J, Joseph D, Thompson T, Wu XC, et al. Recent trends in prostate cancer testing and incidence among men under age of 50. Cancer Epidemiol 2012;36:122-127.ArticlePubMed
- 11. Krajewski KM, Giardino AA, Zukotynski K, Van den Abbeele AD, Pedrosa I. Imaging in renal cell carcinoma. Hematol Oncol Clin North Am 2011;25:687-715.ArticlePubMed
- 12. Rabjerg M, Mikkelsen MN, Walter S, Marcussen N. Incidental renal neoplasms: is there a need for routine screening? A Danish single-center epidemiological study. APMIS 2014;122:708-714.ArticlePubMed
- 13. Znaor A, Lortet-Tieulent J, Laversanne M, Jemal A, Bray F. International variations and trends in renal cell carcinoma incidence and mortality. Eur Urol 2015;67:519-530.ArticlePubMed
- 14. Leveridge MJ, Bostrom PJ, Koulouris G, Finelli A, Lawrentschuk N. Imaging renal cell carcinoma with ultrasonography, CT and MRI. Nat Rev Urol 2010;7:311-325.ArticlePubMed
- 15. Tyson MD, Humphreys MR, Parker AS, Thiel DD, Joseph RW, Andrews PE, et al. Age-period-cohort analysis of renal cell carcinoma in United States adults. Urology 2013;82:43-47.ArticlePubMed
- 16. Cairns P. Renal cell carcinoma. Cancer Biomark 2010;9:461-473.ArticlePubMedPMC
- 17. Suh M, Choi KS, Lee YY, Jun JK. Trends in cancer screening rates among Korean men and women: results from the Korean National Cancer Screening Survey, 2004-2012. Cancer Res Treat 2013;45:86-94.ArticlePubMedPMCPDF
- 18. Etzioni R, Gulati R, Mallinger L, Mandelblatt J. Influence of study features and methods on overdiagnosis estimates in breast and prostate cancer screening. Ann Intern Med 2013;158:831-838.ArticlePubMedPMC
- 19. Zhu Y, Wang HK, Qu YY, Ye DW. Prostate cancer in East Asia: evolving trend over the last decade. Asian J Androl 2015;17:48-57.ArticlePubMedPMC
- 20. Bae JM. Global trends in the use of nationwide big data for solving healthcare problems. J Korean Med Assoc 2014;57:386-390 (Korean).Article
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- Twenty-two-year incidence trend of urological cancers in the Republic of Korea: 1999–2020
Seunghyeon Cho, Won-Ju Park
Investigative and Clinical Urology.2024; 65(1): 23. CrossRef - What is the relationship between the local population change and cancer incidence in patients with dyslipidemia: Evidence of the impact of local extinction in Korea
Wonjeong Jeong, Dong‐Woo Choi, Woorim Kim, Kyu‐Tae Han
Cancer Medicine.2024;[Epub] CrossRef - A single-center long-term experience of active surveillance for prostate cancer: 15 years of follow-up
Sang Hun Song, Jung Kwon Kim, Hakmin Lee, Sangchul Lee, Sung Kyu Hong, Seok-Soo Byun
Investigative and Clinical Urology.2021; 62(1): 32. CrossRef - Risk of thyroid as a first or second primary cancer. A population‐based study in Italy, 1998–2012
Emanuele Crocetti, Veronica Mattioli, Carlotta Buzzoni, Silvia Franceschi, Diego Serraino, Salvatore Vaccarella, Stefano Ferretti, Susanna Busco, Ugo Fedeli, Massimo Varvarà, Fabio Falcini, Manuel Zorzi, Giuliano Carrozzi, Walter Mazzucco, Cinzia Gasparot
Cancer Medicine.2021; 10(19): 6855. CrossRef - Cancer Incidence by Occupation in Korea: Longitudinal Analysis of a Nationwide Cohort
Hye-Eun Lee, Masayoshi Zaitsu, Eun-A Kim, Ichiro Kawachi
Safety and Health at Work.2020; 11(1): 41. CrossRef - The Overdiagnosis of Kidney Cancer in Koreans and the Active Surveillance on Small Renal Mass
Byung Hoon Chi, In Ho Chang
The Korean Journal of Urological Oncology.2018; 16(1): 15. CrossRef - Potential Epigenetic Biomarkers for Prostate Cancer Screening
Jong-Myon Bae
International Neurourology Journal.2018; 22(2): 142. CrossRef - Strategies for Appropriate Patient-centered Care to Decrease the Nationwide Cost of Cancers in Korea
Jong-Myon Bae
Journal of Preventive Medicine and Public Health.2017; 50(4): 217. CrossRef - It needs adaptation to the 2015 Korean guideline for breast cancer screening
Jong-Myon Bae
Journal of the Korean Medical Association.2015; 58(9): 833. CrossRef - Two different views of interpreting the related evidences on the reply to reader's letter
Jong-Myon Bae
Journal of the Korean Medical Association.2015; 58(12): 1196. CrossRef

 KSE
KSE
 PubReader
PubReader ePub Link
ePub Link Cite
Cite